
Practice, not luck, will help you score big on the MCAT. Try your hand at the MCAT sample questions below. Then, check your answers against our in-depth explanations to see how you did.
We pulled these practice questions from our MCAT Subject Review book series and from our MCAT test prep course materials. You can also take a free MCAT practice test with us held under the same testing conditions as the real MCAT. Find out how you'd score, and get a personalized score report from us that shows your strengths and weaknesses.
One difficulty in following Adam Smith’s account of self interest is that he had discussed the matter thoroughly in the Theory of Moral Sentiments , and he assumed that the reader of the Wealth of Nations would not think that he, Smith, considered self-interest the only or even the main motive, or virtue, of humanity. His teacher, Hutchenson, indeed, had taught that the only virtue was benevolence; but Smith, while agreeing that this was the major virtue and the one which aimed “at the greatest possible good,” felt strongly that the system of benevolent ethics was too simple and left no room for the “inferior virtues.” Therefore he devoted himself to a more naturalistic theory of morals, in which man’s nature was accepted as it was.
In the Wealth of Nations , Smith combined the two doctrines: God’s providential benevolence and man’s earthly self-interest. The result is his famous “invisible hand” theory in which the individual, intending only his own gain, is led “to promote an end which was no part of his intention,” the well-being of society. The view that personal self-interest is the best regulator of public affairs had been put forward before: it is expressed in Bernard de Mandeville’s, Private Vices, Public Benefits . When Smith wrote, this view was already familiar to eighteenth century thinkers. What Smith did was to give it a reasoned economic exposition which made it acceptable and, so to speak, respectable. From then on, the inevitable benefits of self interest become a doctrine to which rising manufacturers and owners of newly enclosed land constantly appealed. However, he was constantly inveighing against the farmers, the workers, the manufacturers, and the banks, complaining that they did not understand their own particular interests. He chided the mercantilists that their very cupidity, by imposing a heavy duty on certain goods, called into being a smuggling of the goods which ruined their business. Country gentlemen were told that in their demand for a bounty on corn “they did not act with that complete comprehension of their own interest” which should have directed their efforts.
Smith’s method was to form out of experience an abstract principle, to state this as a general rule and to give evidence and examples to support it. Thus, he and his science of economics could show “how” and “in what manner.” In order to discover such a science of economics, however, Smith had to posit a faith in the orderly structure of nature, underlying appearances and accessible to man’s reason. This, in our judgment, is what Smith really meant by the “invisible hand”; that, so to speak, an “order of nature” or a “structure of things” existed which permitted self-interest, if enlightened, to work for mankind’s good.
Man’s task, therefore, was to understand the nature or structure of things and to adjust himself harmoniously to the necessary results of this structure. On one level, this might mean the acceptance of a “natural” price of things (reached when the supply, whether of goods or of labor, exactly equaled the demand). On another level, Smith applied his faith in a structure of things when he said: “A nation of hunters can never be formidable to the civilized nations in their neighbourhood. A nation of shepherds may.” This is true, he thought, because the nature of hunting is such that large numbers cannot indulge in it; the game would be exterminated. On the other hand, shepherds can grow in number as their flocks grow: and can carry war into the hearts of civilized nations because they carry with them their food supply.
What effect did Smith’s work actually have? First, it gave the rising manufacturers and merchants a rationale for their desire to change existing government policy. (Existing policy, as we have pointed out, favored the older trades, methods, and classes against the new “Lunar Society” type of individual and enterprise.) Thus, for example, it helped Pitt to pass a free-trade agreement, the Eden Treaty of 1786 with France, through Parliament.
The second effect of Smith’s work was in the shaping of thought. His influence in introducing historical method into political economy was far-reaching. He made the foundation of all subsequent economics the notion that wealth was created by labor. But, more than any of these things, he introduced science into the study of economics. Although he talked much about the “invisible hand” and the “natural course of things,” Smith really freed man from the tyranny of chance by forming for him the analytical tools with which he might learn to control his economic activities.
Material used in this particular passage has been adapted from the following source: J. Bronowski and B. Mazlish, The Western Intellectual Tradition . ©1960 by HarperCollins Publishers.
1. Which of the following statements, if true, would most undermine the author’s characterization of Smith?
(A) Smith extrapolated his theories from real-life observations.
(B) Smith’s work was wholly theoretical.
(C) Smith based part of his work on an older idea.
(D) Smith’s theory influenced the work of later economists.
Answer: (B)
A: No. The author characterizes Smith as forming an abstract principle out of real-life experience (paragraph 3.) Therefore, this choice is consistent, not inconsistent, with the passage.
B: Yes. The author stresses the real-life evidence formulating Smith’s theories, and the real-life impact they had (see paragraph 2 for examples). Therefore this statement, if true, would undermine the author’s characterization of Smith.
C: No. Smith did base his work in part on the work of Hutchenson and de Mandeville (see paragraphs 1 and 2). This choice is consistent with the passage.
D: No. The author states that Smith’s work laid the foundation of all subsequent economic studies (see paragraph 6). This choice is consistent with the passage.
2. Which of the following items of information from the passage most supports the author’s claim that Smith believed that the economy can be not only studied but influenced by human actions?
(A) Smith felt that the system of benevolent ethics was too simple and left no room for the “inferior virtues.”
(B) Smith combined two doctrines to create his “invisible hand” theory.
(C) Smith introduced a historical method into the study of economy.
(D) Smith criticized businessmen who did not act in their own best interest.
Answer: (D)
Note: This question asks you to decide which of the four statements cited in the choices is most directly used within the passage as support for the author’s own claim that Smith believed the economy can be influenced.
A: No. This statement represents the inspiration for Smith’s theory, but does not demonstrate that Smith feels that the economy can be influenced.
B: No. The fact that the “invisible hand” theory combines two different elements (the doctrines of “God’s providential benevolence and man’s earthly self-interest” (paragraph 2) does not show that Smith feels that the economy can be influenced. These are two separate issues in the passage.
C: No. This answer speaks to how the economy is now studied (paragraph 6), but has no direct relevance to whether or not the economy can actually be influenced.
D: Yes. The fact that Smith actively tried to get farmers, workers, and manufacturers to act in their own best interest (and not just their perception of it) clearly indicates that by changing their behavior, he feels that he can change the marketplace (see paragraph 2). Therefore, out of the four choices (all of which are from the passage), this information most acts to give support for the claim cited in the question stem.
3. According to the author, Smith’s most important contribution to economics was:
(A) identifying benevolence as man’s only virtue. Answer: (D)
A: No. This was Hutchenson, not Smith (see paragraph 1).
B: No. Smith used the term “nature” but the passage does not suggest that he was the first to connect human nature (or the nature of things in general) to economics, or that this was the most important aspect of Smith’s work.
C: No. This was an aspect of his contribution, but the author specifically describes the “science” that Smith provides as his most important contribution to the study of economics (paragraph 6).
D: Yes. The author stresses that his lasting and most important contribution was introducing a scientific methodology to economics (paragraph 6).
Mammalian nerve cells have on their outer surface a subtype of glutamate receptors called the N-methyl-D-aspartate (NMDA) receptor. The NMDA receptor binds glutamate, an amino acid neurotransmitter, which ultimately results in the inward flow of calcium ions.
The NMDA receptor has been studied by exposing nerve cells to ischemic conditions (a diminished flow of blood) which result in localized brain damage. The affected neurons demonstrate depleted energy reserves with decreased internal stores of ATP. Energy-driven Na + /K + ATPase enzymes located in the cell membrane begin to fail. If the ischemic conditions continue, the neuronal cell membrane depolarizes, providing an excitatory stimulus for the release of excessive amounts of glutamate. Other nerve cells in close proximity experience sustained binding of glutamate to NMDA receptor sites, which can lead to further cell membrane depolarization.
Cultured nerve cells were exposed to normal or decreased oxygen concentrations. Differing concentrations of extracellular calcium or glutamate antagonists were also established. Intracellular calcium levels were measured after one minute. The results of this experiment are listed below:
| Extracellular environment | Intracellular calcium | ||
|---|---|---|---|
| Oxygen | Glutamate antagonists | Calcium | |
| L | − | − | N |
| L | − | H | I |
| L | + | − | N |
| L | + | H | N |
| H | − | − | N |
| H | − | H | N |
| H | + | − | N |
| H | + | H | N |
Key:
Table 1
Several cultured neurons were bathed in a solution containing high concentrations of calcium, sodium, and chloride ions, but not oxygen. The cells experienced marked swelling and eventually an action potential was evoked. After the action potential, internal ion levels were measured every 30 seconds. Intracellular levels of both calcium and sodium were elevated.
Based on these two experiments, researchers have proposed that the initial effects of activation of the NMDA receptor can be modeled as follows:
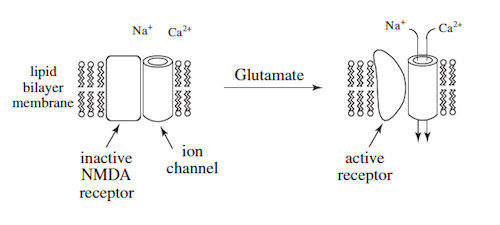
Figure 1
Further experiments suggested that the NMDA receptor also allowed K + to exit the cell.
1. Regarding Experiment 2, which of the following is the most likely reason that the cells experienced marked swelling?
(A) Activation of the NMDA receptor due to ischemia blocked aquaporins, preventing the efflux of water from the cell.
(B) The failure of the Na + /K + ATPase led to osmotic influx of water.
(C) The high intracellular concentrations of sodium and calcium led to the osmotic influx of water.
(D) The influx of sodium due to the action potential led to the osmotic influx of water.
Answer: (B)
The cells were kept in ischemic (low oxygen) conditions. Oxygen is required for ATP synthesis, and ATP is required to operate the Na + /K + ATPase. In the absence of oxygen, the lack of ATP would cause the Na + /K + ATPases to fail. One of the functions of the ATPases is to maintain osmotic equilibrium for the cell; as it pumps ions out, water doesn’t want to come in. The failure of the pump would lead to osmotic influx of water (choice B is correct). There is nothing in the passage to support the idea that NMDA receptors block aquaporins, and even if they did, the movement of water through the aquaporins is not restricted to just efflux (choice A is wrong). While the increase in intracellular concentrations of sodium and calcium could lead to osmotic influx of water, this would happen after the action potential was induced, and the swelling occurred prior to the induction of the action potential (choices C and D are wrong).
2. Which of the following amino acids would most likely be found in the transmembrane portion of the NMDA receptor?
(A) Leu
(B) Pro
(C) Arg
(D) Asn
Answer: (A)
Proteins that span the plasma membrane must contain hydrophobic (nonpolar) amino acid residues in the transmembrane region. Of the amino acids listed, only leucine and proline are nonpolar; arginine and asparagine are both polar (choices C and D can be eliminated). Proline, while nonpolar, does not fit well into the alpha helical shape of most transmembrane regions, making leucine the more likely of the two to be found there (choice B can be eliminated, and choice A is correct).
3. Regarding Experiment 1, if glutamate is the only neurotransmitter that can bind to the NMDA receptor to increase the inward flow of calcium, which of the following would account for the lack of calcium flow in the presence of oxygen, extracellular calcium, and no glutamate antagonists?
(A) No glutamate was supplied to the cells in the extracellular medium.
(B) Other glutamate receptor subtypes inhibit the flow of calcium into the cell.
(C) In the presence of oxygen the cell does not depolarize.
(D) Ischemic conditions prevent the normal functioning of the Na + /K + ATPases.
Answer: (C)
In the presence of oxygen, the Na + /K + ATPases would not fail, and the cell would not depolarize. If the cell doesn’t depolarize, then glutamate would not be released to stimulate the NMDA receptor and open the calcium channels (choice C is correct). It is true that no glutamate was supplied to the cells, but this was by design; the experiment was designed to test what happens with glutamate release and NMDA receptor stimulation in the absence of oxygen. Supplying glutamate in the extracellular medium would prevent any effect of ischemia from being seen since the cell would automatically be stimulated (choice A is wrong). It is also true that ischemic conditions prevent the normal activity of the ATPases, but these cells are not exposed to ischemic conditions (choice D is wrong), There is no reason to assume any other glutamate receptor subtypes (choice B can be eliminated).
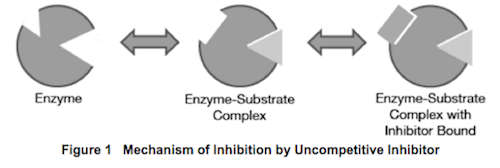
Inhibition of enzymes by various biological and non-biological substrates falls into one of four basic categories: competitive, noncompetitive, uncompetitive and mixed inhibition. Unlike either competitive or non-competitive inhibition, uncompetitive inhibition results when the inhibitor binds specifically to the enzyme-substrate complex as opposed to the unbound enzyme. One possible mechanism explaining this behavior is a change in the shape of a pocket on the enzyme that is separate from the active site upon substrate binding. This induced change allows a molecule to bind in the pocket, which would otherwise be unavailable in the absence of the substrate, and the binding of that molecule inactivates the complex. This mechanism is demonstrated in Figure 1 below.
The process by which this occurs can be described mathematically by modifying the Michaelis-Menten equation:
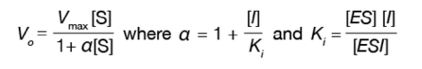
where V 0 is the initial velocity, V max is th emaximum velocity, [S] is the concentration of substrate, [I] is the concentration of inhibitor, K i is the dissociation constant for the inhibitor and enzyme-substrate complex binding, [ES] is the concentration of the enzyme-substrate complex, and [ESI] is the concentration of the enzyme-substrate complex with inhibitor bound. When 1/V 0 is plotted against 1/ [S| at various [I] , parallel lines result, which are shifted upwards with increasing [I] .
Cobalamin-dependent methionine synthase (MetS) is a transmethylase enzyme that utilizes a methylcobalamin (Me-CbI) cofactor and plays a critical role in methionine metabolism. MetS uses the Me-CbI cofactor to transfer a methyl group derived from methyl tetrahydrofolate (MTHF) to homocysteine to form methionine and tetrahydrofolate (THF), which is required for the synthesis of purines and pyrimidines. In order to investigate the effect of various inhibitors on MetS, a recent study prepared MTHF analogs to determine their effect on the enzyme’s overall rate. The results of three of these inhibitors, 5c, 3g, and 3j are shown below in Figure 2.

Adapted from Bioorganic & Medicinal Chemistry (2014) 22, pages 550-558. Published online 8 November 2013.
1. According to the passage, which of the following plays a role in uncompetitive inhibition?
I. Inhibitor binding to the free enzyme
II. Substrate analog binding in the active site
III. Change in the shape of the inhibitor binding site
Answer: (B)
Because all the items appear twice in the answer choices, there is no best item to examine first, so we will begin with item I. According to the passage, uncompetitive inhibitors only bind to the enzyme-substrate complex, making item I incorrect (eliminate answer choices A and C). Item II describes the process by which competitive inhibition occurs. In uncompetitive inhibition, the inhibitor may resemble the substrate but it binds at a location separate from the active site (Item II is false; choices C and D are incorrect). Item III is described in the passage and results from substrate binding (answer choice B is correct).
2. How does uncompetitive inhibition affect the apparent K m and the V max of a given enzyme?
Answer: (D)
Binding of the uncompetitive inhibitor will effectively decrease the concentration of enzyme available for catalysis, thus decreasing V max (answer choices A and C are incorrect). Because the inhibitor can only bind to the enzyme-substrate complex, it increases the apparent affinity of the enzyme for its substrate, thus decreasing apparent K m (answer choice D is correct; choice C is wrong). The passage states that uncompetitive inhibition results in sets of parallel lines that are shifted upwards with increasing concentration of inhibitor when 1/V 0 is plotted against a 1/ [S] (a Lineweaver-Burk Plot). Given that slopes of parallel lines are the same and considering that the slope of the line on a Lineweaver-Burk Plot is K m /V max then both K m and V max must be affected equally because the slope is unchanged. If you did not recall the effect of an uncompetitive inhibitor on V max and apparent K m from memory, you could refer to Figure 2. Examining graph 3j, we see that with increasing inhibitor concentration, the y-intercept (corresponding to 1/V max increases and the x-intercept (corresponding to −1/K m becomes more negative. Given that 1/V max y-intercept) is getting larger, V max must be getting smaller due to the inverse relationship. The value of K m is slightly more confusing, so we will use some numbers to demonstrate the direct relationship. For an x-intercept of −2: K m = −1 ⁄ −2 =½.For an x-intercept of −4; K m = −1 ⁄ −4 =¼.Thus, as the x-intercept decreases, the value of K m also decreases.
3. According to information presented in the passage, what type of inhibitors were the researchers attempting to synthesize?
Answer: (A)
According to the passage, the study aimed to create MTHF analogs, and MTHF is one of the substrates of MetS. Inhibitors that are designed to mimic the shape of a substrate are competitive inhibitors, which competitively bind to the active site (answer choice A is correct). Noncompetitive and uncompetitive inhibitors do not need to be substrate analogs because they do not bind the active site, but instead bind to a separate site on the enzyme, an allosteric site (answer choices B and C are incorrect). Mixed inhibitors may exhibit a combination of competitive and noncompetitive inhibition, but competitive inhibitors are always substrate analogs by definition, so answer choice A is the best answer (choice D is wrong).
Medical and law schools socialize their students to fit into the professional roles of physicians and lawyers. During this socialization process, students face major conflicts that can lead to negative outcomes. The stress that medical and law students face at school can also result in strain among the students’ interpersonal relationships.
Researchers discovered that women are more likely to experience stress than men during their training periods because of role strain: the built-in conflict that results from the woman having to choose between the demands placed on her by her profession and those that stem from her obligations as a woman/mother/wife and from her identity as a female. Women in professional programs are still, to some extent, considered “tokens” (members of a minority group representing the whole group, rather than individuals representing only themselves), because professional schools predominantly historically enrolled mostly male students. Women who describe themselves as having masculine personality traits reported having more stress in these professional schools than those who believed they had feminine traits.
Researchers designed a study to observe patterns of stress among students in professional schools. They predicted that most students’ stress originates from interpersonal relationships rather than from environmental or structural factors. They also predicted that more women than men would report stress from gender discrimination because these professions are male-dominated. For this study, 400 first and second year medical and law students (25% women and 75% men) were given a survey with questions that measured four main factors: program stressors (the levels and sources of stress), impaired relationships (the impact of medical/law schools on relationships with friends, family, and spouses), psychological stress (changes in behavior due to mental and emotional anxieties), and role obligation (roles that students presumed themselves to have). Figure 1 shows the results of the questions about sources and degrees of stress.
| Degree of Stress | ||||||
| Source of Stress | Very Stressful | Moderately Stressful | Not Very Stressful | |||
| Male | Female | Male | Female | Male | Female | |
| Financial pressures | 12.2 | 15.7 | 43.6 | 46.0 | 44.2 | 38.3 |
| Interaction with faculty members | 3.3 | 4.4 | 27.4 | 24.8 | 69.3 | 70.8 |
| Difficulties with spouse/partner | 8.8 | 12.4 | 24.8 | 36.5 | 66.4 | 51.1 |
| Sexism | 1.2 | 8.4 | 12.9 | 58.1 | 85.9 | 33.5 |
| Racism | 2.8 | 1.7 | 5.8 | 10.7 | 91.4 | 87.6 |
| Shortage of time | 39.0 | 38.2 | 55.6 | 58.1 | 5.4 | 3.7 |
| Deadlines | 48.1 | 52.7 | 40.7 | 41.6 | 11.2 | 5.7 |
| Examinations | 57.4 | 57.9 | 30.2 | 28.9 | 12.4 | 13.2 |
| Competition with classmates | 25.3 | 29.9 | 29.4 | 23.7 | 45.3 | 46.4 |
Figure 1. Results of students’ responses reporting the sources and degrees of stress after enrolling in medical or law school (as percentage)
Adapted from E. J. Clark & P. P. Rieker. Gender differences in relationships and stress of medical and law students. © 1986 by Journal of Medical Education
1. Which of the following statements can be inferred about role strain for women?
I. Women tend to be under more stress than men in professional schools due to impression management.
II. A female student in medical school is an example of front-stage self.
III. A mother interacting with other mothers at the park is an example of back-stage self.
(A) I and II only
(B) I and III only
(C) II and III only
(D) I, II, and III
Answer: (A)
Item I is true: Since role strain is conflict resulting from having multiple identities, or playing multiple roles, it is related to impression management: people use impression management to craft their front-stage selves, and the difficulty of this can cause stress, known as role strain (choice C can be eliminated). Item II is true: A front-stage self in the dramaturgical perspective is the self the individual “performs” in front of an audience: the female’s role as a student in medical school is performed in front of other members of society such as the other medical students, teachers, and staff (choice B can be eliminated). Item III is false: A back-stage self in the dramaturgical perspective is the self the individual is not performing in front of any audience: the woman in this scenario is still playing the role of the mother in front of other mothers, which is considered front-stage behavior (choice D can be eliminated and choice A is, therefore, the correct answer).
2. Suppose most people believed male students were more likely to excel in medical school than female students. Which of the following illustrates stereotype threat for this scenario?
(A) Female students producing low exam scores due to anxieties about performing poorly.
(B) Male students producing high exam scores due to anxieties about performing poorly.
(C) A majority of women not applying to medical school because they are not interested in the science field.
(D) A majority of men applying to medical school because they are pressured by others to pursue careers in medicine.
Answer: (A)
Stereotype threat occurs when an individual’s performance is affected by feeling anxious about being labeled with a negative stereotype. The female students perform worse on exams due to anxiety about the negative stereotype (choice A is correct). The male students do not perform poorly because of a negative stereotype; in fact, the reverse happens (choice B is wrong). Women who do not choose to be in medical school because of their lack of interest are not responding to stereotype threat (choice C is wrong). Men who feel apply to medical school because of pressure from others are responding to peer pressure rather than stereotype threat (choice D is wrong).
3. Do the results of the study accurately support both the researchers’ hypotheses?
(A) The results support both hypotheses because Figure 1 shows that students experience lower amounts of stress due to interpersonal relationships than due to structural factors.
(B) The results support both hypotheses because Figure 1 shows that women reported more stress from sexism than did men.
(C) The results do not support both hypotheses because Figure 1 shows that men reported more stress due to racism than did women.
(D) The results do not support both hypotheses because Figure 1 shows that students experience higher amounts of stress due to structural factors than due to interpersonal relationships.
Answer: (D)
The hypotheses mentioned in the passage are that “most students’ stress originates more from interpersonal relationships than from environmental and structural factors” and that “more women than men would report stress from gender discrimination because these professions are male-dominated.” While the second hypothesis is borne out by the data (women face more stress than men due to sexism), Figure 1 also shows that structural factors (exams, deadlines, and shortage of time) caused students more stress than interpersonal relationships with faculty, spouses, and peers; the figure does not support both hypotheses (choice D is the correct answer). The results do not fully support both hypotheses of the study (choice A and choice B are wrong). Gender discrimination is equivalent to sexism, not racism; racism is unrelated to the study’s hypotheses (choice C is wrong).
Blood pH homeostasis is the result of several systems operating within the bloodstream. They collectively maintain blood plasma pH at 7.4, since a drop in pH below 6.8 or rise above 7.8 may result in death.
One component of this system is the enzyme carbonic anydrase , which catalyzes the conversion of CO 2 in the blood to carbonic acid. Carbonic acid, in turn, ionizes to form the carbonic acid-bicarbonate buffer. The interdependence of these reactions is shown below in Equation 1.

Equation 1
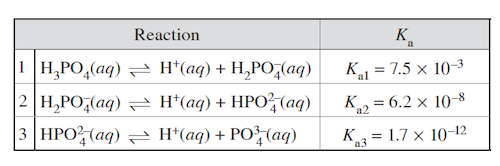
Uncatalyzed blood CO 2 i and H + can be found binding to hemoglobin after oxygen liberation in peripheral tissues. As the blood reaches the lungs these actions reverse themselves; hemoglobin binds with oxygen, releasing the CO 2 i and H + ions. The exchange of gases between the lungs and the blood and other tissues in the body is a physiologic process known as respiration. A second system, the phosphoric acid buffer, plays a minor role compared to the carbonic acid-bicarbonate buffer. Phosphoric acid (H 3 PO 4 ), the primary reactant of this system, is a triprotic acid, which can ionize three protons. This three-step process is illustrated below:
1. If CO 2 gas is bubbled continuously in a beaker of water to form carbonic acid, which of the following would be true?
(A) I only
(B) II only
(C) III only
(D) II and III
Answer: (D)
Addition of a catalyst (such as the enzyme carbonic anhydrase) will simply increase the rate of a reaction. It plays no role in shifting the equilibrium, or changing the equilibrium constant making Item I false (eliminate choice A). As carbon dioxide is bubbled through, carbonic acid will form until its equilibrium concentration is attained, making Item II valid (eliminate choice C). Finally, addition of bicarbonate will shift the carbonic acid equilibrium to the reactant side, consuming H+ in the process. Since the concentration of H + will decrease, pH will increase, making Item III valid (eliminate choice B).
2. In the dissociation of phosphoric acid, the trend K a1 > K a2 > K a3 is predominantly due to:
(A) an equilibrium shift towards the reactants side in Reactions 2 and 3 due to the release of H + in Reaction 1.
(B) a smaller radius in the H + liberated in Reaction 1 compared to that in Reactions 2 and 3.
(C) a slower rate of reaction after subsequent ionizations.
(E) an increasing influence of the anion after subsequent ionizations.
Answer: (D)
Generation of H + in Reaction 1 is coupled with a release of H 2 PO 4 − . Both the product and reactant sides of Reaction 2 are increased proportionally, causing no shift in equilibrium (eliminate choice A). Atomic radius is a function of an atom’s position in the periodic table. Thus, the radius of H + is the same in all three reactions, eliminating choice B. Equilibrium constants have no relationship to reaction rates, so choice C can be eliminated. The K a values progressively decrease when removing a proton from a polyprotic acid because it is more difficult to remove a proton from an anion compared to a neutral molecule. In subsequent ionizations, the anion becomes more negative, resulting in greater difficulty liberating a positively charged H + ion.
3. What would be the pH of a solution made from combining 50 mL of 0.030 M acetic acid (K a = 1.8 × 10 −5 ) and 10 ML of 0.15 M sodium acetate?
(A) pH = 2.5
(B) pH = 2.5
(C) pH = 3.3
(D) pH = 4.7
Answer: (D)
The final solution is composed of (50 mL)(0.03 M) = mmol of HC 2 H 3 O 2 and (10 mL)(0.15 M) = 1/5 mmol of NaC 2 H 3 0 2 (or 1.5 mmol of C 2 H 3 O 2 − ). The total volume will be 60 mL and the starting concentration of acetic acid will be the same as the starting concentration of its conjugate base. Since acetic acid is a weak acid, any subsequent dissociation will be relatively insignificant and the equilibrium concentrations of acid and base will remain approximately the same. When the concentration of the two species in a conjugate pair are equal, the pK a = pH the Henderson-Hasselbalch equation: pH = pK a + log [conjugate base]/[acid]. The pK a of acetic acid (K a = 1.8 × 10 −5 ) is approximately 4.7.
Bile is a greenish fluid produced in the liver that helps dispose of the liver’s waste products and aids in the digestion of fats. It is stored in the gallbladder and emptied into the small intestine via the common bile duct when needed. The principle components of bile are bile acids, cholesterol, and bilirubin. The liver enzymatically converts cholesterol (1) into one of two primary bile acids, cholic acid (2) or chenodeoxycholic acid (3).
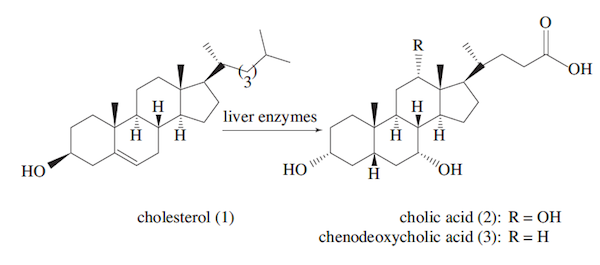
These acids are then coupled with glycine, an amino acid, or taurine, one of the few known naturally occurring sulfonic acids. Taurocholic acid (4) is readily converted into its salt in the duodenum upon mixing with pancreatic secretions containing lipase and bicarbonate.
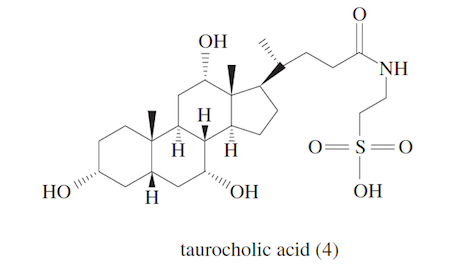
Bile acids are amphipathic. It is this property that allows them to emulsify fat globules into microscopic micelles, increasing fat surface area and aiding digestion by lipase.
Bacteria in the colon convert Compounds 2 and 3 into their respective secondary bile acids 5 and 6, shown below. Bile salts are not passively absorbed in the small intestine, but secondary bile acids are actively absorbed in the colon where the pH is less basic. Up to 95% of the bile acids produced by the liver are reabsorbed and can be used in the digestive process up to twenty times.
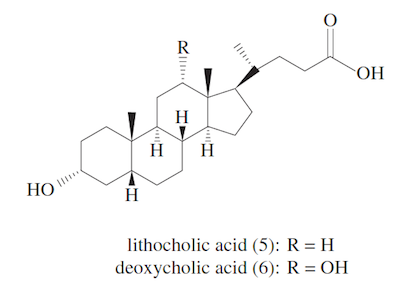
1. Which of the following contributes to the fact that the small intestine cannot passively absorb bile salts?
(A) I only
(B) I and II only
(C) I and III only
(D) II and III only
Answer: (C)
Salts are charged, making them water soluble or hydrophilic; Item II is false (choices B and D can be eliminated). By looking at the remaining choices, Item I must be true, so focus on Item III to answer the question quickly. The passage states that pancreatic secretions contain bicarbonate, a base, which raises the pH of the small intestine. High pH means a high concentration of OH − is present to deprotonate the bile acid, yielding its charged conjugate base. Since Item III is a true statement, choice C is the best answer. To address Item I, the pK a of a functional group is smaller than the environmental pH, the group will exist in its deprotonated or charged form. This confirms Item I as true.
2. Lipase works to hydrolyze which of the following functional groups?
(A) Amide
(B) Ester
(C) Ether
(E) Hemiacetal
Answer: (B)
According to the passage, lipase is an enzyme that breaks down fats, or triglycerides, which contain three ester functional groups. Amides are the functional groups present in proteins so eliminate choice A, hemiacetals are present in sugars (eliminate choice D), and ethers are not largely important functional groups in any biologically important molecules (eliminate choice C).
3. Taurine is a derivative of the amino acid cysteine, shown below. Which of the following reactions is a necessary part of the biochemical conversion of cysteine to taurine in the body?
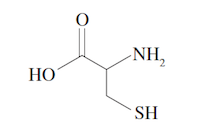
(A) Decarboxylation of the carboxyl group
(B) Protonation of the basic amine
(C) Reduction of the thiol group
(D) Conversion of the primary amine to an imine
Answer: (A)
Compare the structure of cysteine given in the question to the structure of taurine, which can be deduced from the structure of taurocholic acid given in the passage. 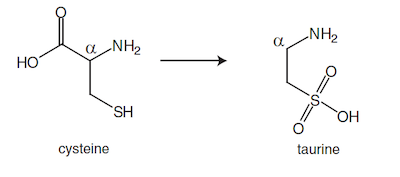 The amino group is still present in taurine and has been coupled to the carboxyl group of the bile acid to form a new amide bond (eliminate choices B and D). The thiol group in cysteine cannot be reduced anymore, but has instead been highly oxidized with the addition of five bonds to oxygen in order to form taurine (eliminate choice C). By process of elimination, decarboxylation must have taken place. The COOH group of the amino acid has been lost since no carbonyl group is present in the new substituent of Compound 4 in the passage.
The amino group is still present in taurine and has been coupled to the carboxyl group of the bile acid to form a new amide bond (eliminate choices B and D). The thiol group in cysteine cannot be reduced anymore, but has instead been highly oxidized with the addition of five bonds to oxygen in order to form taurine (eliminate choice C). By process of elimination, decarboxylation must have taken place. The COOH group of the amino acid has been lost since no carbonyl group is present in the new substituent of Compound 4 in the passage.
Students are performing experiments in the laboratory using their knowledge of hydrostatics and hydrodynamics.
Students are given five liquid substances and they are asked to find their densities. They also have a test block that they measure to have a mass of 50 g and a volume of 100 cm3. They place the test block into each liquid and measure how much of the test block is submerged in the water. Table 1 summarizes their results.
| Liquid | Volume submerged | Float? |
|---|---|---|
| 1 | 80 cm 3 | Yes |
| 2 | 75 cm 3 | Yes |
| 3 | 100 cm 3 | No |
| 4 | 50 cm 3 | Yes |
| 5 | 100 cm 3 | Yes |
Table 1: Test block placed in different liquids
The students are given four different complex objects that all have the same density. They place each object in a test liquid that has a specific gravity of 2. They record the submerged volume of the object by the measuring the displacement of the liquid. Their results are summarized in Table 2.
| Object | Volume of displaced liquid |
|---|---|
| A | 150 cm 3 |
| B | 90 cm 3 |
| C | 75 cm 3 |
| D | 100 cm 3 |
Table 2: Complex objects in a test liquid
Students must create an irrigation system that takes water from a reservoir 80 cm deep to a wave pool across the room. A perfectly leveled, horizontal tube with constant circumference takes water from the bottom of the reservoir to the wave pool.
1. From Experiment 1, which liquid has the smallest density?
(A) Liquid 4
(B) Liquid 3
(C) Liquid 5
(D) Liquids 3 and 5
Answer: (B)
An object will only sink if its density is greater than the density of the liquid. Since the test object is the same for all liquids, and only sank in Liquid 3, then Liquid 3 has the smallest density.
2. If the tubing used in Experiment 3 has a cross-sectional area of 0.02 m 2 , what is the velocity of the water as it enters the wave pool? Assume the tubing is soft so that the pressure at the aperture from the reservoir is P atm .
(A) 2 m/s
(B) 3 m/s
(C) 4 m/s
(D) 5 m/s
Answer: (C)
The velocity of the water entering the tube is given by v = (2gD) 0.5 = 4 m/s. The flow rate is constant, and the cross-sectional area of the tubing does not change, so the velocity at the wave pool is equal to the velocity as it exits the reservoir.
3. Two objects made from the same material with the same mass are placed in a liquid, base first. The base of Object 2 is three times that of Object 1. What best describes the buoyant force on the objects?
(A) Object 1 has a greater buoyant force acting on it because it has a larger volume submerged.
(B) Object 2 has a greater buoyant force acting on it because it has a larger volume submerged.
(C) Object 2 has a greater buoyant force acting on it because it has a larger area at its base.
(D) The buoyant force acting on both objects is the same.
Answer: (D)
The buoyant force is given by the equation: 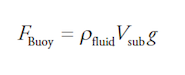
Since the blocks have the same density, they will have the same volume submerged, and thus, they will have the same buoyant force acting on them.
Explore our featured graduate schools & programs to find those that both match your interests and are looking for students like you.
Check out our complete list of 168 law schools, based on surveys of school administrators and over 17,000 students.
Visit our Med School Hub to explore med schools with our ‘Find Your Med School’ filtered search or visit our Med School Advice pages for info about good MCAT scores or interview question prep.

Explore our featured business schools to find those that are looking for students like you.